Should everyone get a blood test for cancer?
Promise, hype, and hard truths
Over the years I’ve done thousands of routine physicals. Often people come in expecting me to give them a clean bill of health that includes screening for any kind of cancer they might have lurking within. When I explain that I’ll do my best, but that there are only a few types of cancer (think colon, breast, cervical, lung, and maybe prostate cancer) for which routine screening actually reduces mortality… there is a perceptible deflation. My reply usually lands with a disappointed thud. Really? But I want to know I have no heart disease or cancer or other stuff. I want to be around for a long time for my family.
I get it.
Now, imagine a single blood test that could look for dozens of cancers at once, long before symptoms appear, and tell where in your body to look. That’s the promise of multi‑cancer early detection (MCED) tests—a new generation of screening tools that have drawn huge excitement, intense marketing (see Super Bowl ads for example), and now, some sobering results.
Two MCED tests are currently marketed in the U.S.:
Galleri, made by GRAIL
CancerGuard, made by Exact Sciences
Both are sold as “laboratory‑developed tests”, meaning they are available by prescription even though they are not yet FDA‑approved. And as of early 2026, neither has been proven to reduce cancer deaths. That is the key context for everything that follows.
This post walks through what these tests do, what the data actually show, what the latest big trial in England found, and how to think about risks and benefits if you’re considering one. I’ve been working on this post since before the Super Bowl, but decided to drop it now, one day after the bombshell announcement by GRAIL yesterday about their clinical trial results.
Would I get one of these tests? Yearly? How will I reply to patients who ask for my opinion about these blood tests? Will I order one for them as requested?
Here are the evidence based details, you know, examined.
Why MCED tests exist at all
We all know by now that traditional cancer screening is disease‑by‑disease: mammograms for breast cancer, colonoscopy or stool tests for colorectal cancer, Pap tests for cervical cancer, PSA for prostate cancer, low‑dose CT scans for lung cancer in high‑risk smokers. These tests save lives, but they cover only a handful of cancers.
Meanwhile:
Most cancer deaths come from cancers that have no routine screening test—like pancreatic, ovarian, liver, esophageal, and some head and neck cancers.
These cancers are often diagnosed at Stage IV, when cure is rare and treatment is mainly about control, not eradication.
MCED tests try to fill that gap. They aim to:
Detect many cancers at once from a blood sample.
Catch at least some of the currently “unscreened” cancers at earlier, more treatable stages.
Potentially complement, not replace, existing single‑cancer screening.
In theory, that could be transformative. In practice, as we just got a glimpse from the GRAIL trial results yesterday, the real picture is more complicated.
Galleri: the flagship MCED test
Galleri looks for fragments of tumor DNA that might be floating in the blood—“cell‑free DNA”—and focuses on methylation patterns, a type of chemical tag on DNA that differs between healthy and cancer cells. Using machine learning, the test tries to answer two questions:
Is there a cancer signal in the blood?
If yes, what is the likely Cancer Signal Origin. For example, pancreas vs ovary vs lung?
If Galleri says “Cancer Signal Detected,” it also reports a predicted tissue or organ of origin, which then guides imaging and other tests. This is a key feature, as just getting a maybe somewhere result is a mess. Read on.
How accurate is it?
There are three key numbers for any screening test:
Sensitivity – how often it finds cancer when cancer is truly present.
Specificity – how often it correctly says “no cancer” when there really is no cancer.
Positive Predictive Value (PPV) – if the test is positive, how likely is it to really be cancer?
From large Galleri studies:
PATHFINDER and PATHFINDER 2 (prospective real‑world screening trials) show:
Sensitivity for cancers caught within 12 months of testing: about 40% for all cancers, ~74% for the 12 deadliest cancers.
Specificity ~99.6%, meaning only ~4 false positives per 1,000 people tested.
PPV ~62%: when Galleri was positive, about 6 in 10 people truly had cancer.
Cancer Signal Origin accuracy around 90%+.
In plain English:
Galleri is very good at avoiding false alarms.
When it says “cancer signal,” it’s more often right than wrong.
But it misses most cancers (100% - 40% for all cancers = 60% missed), especially the earliest ones.
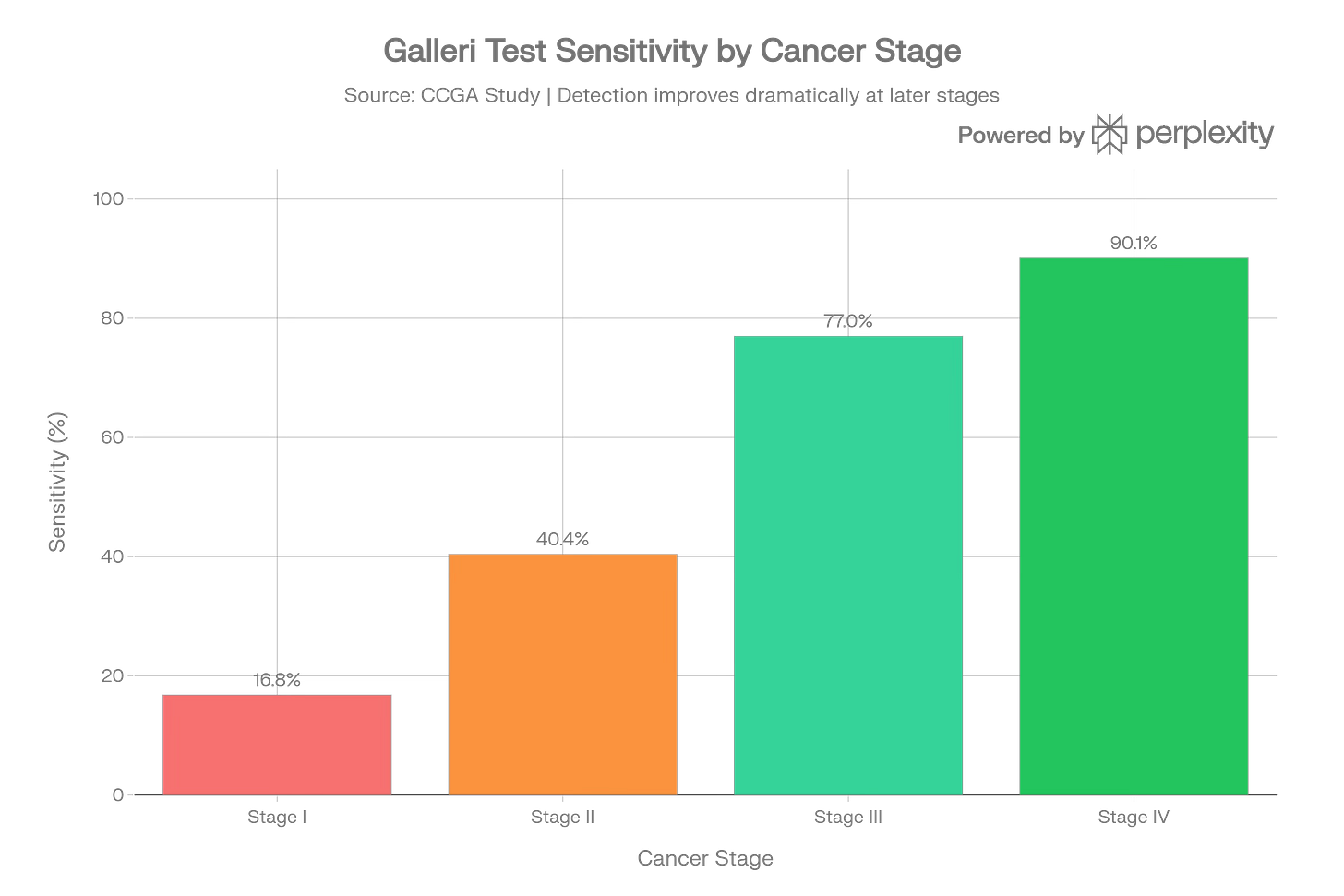
Catching a chance early is obviously the goal of any cancer screening, as earlier detection leads to earlier treatment and better results. How early are cancers found with Galleri? In earlier case‑control data, Galleri’s sensitivity by stage was roughly:
Stage I: ~17%
Stage II: ~40%
Stage III: ~77%
Stage IV: ~90%
So the test is best at finding more advanced cancers and least sensitive at Stage I—the exact opposite of what many people assume. Here is what that looks like, in a graph I made with help from Perplexity:
What that big NHS‑Galleri trial just showed
The NHS‑Galleri trial in England was the high‑stakes test for this technology. It enrolled ~142,000 adults ages 50–77, randomized them to:
Usual National Health System screening
vs.Usual screening plus annual Galleri testing for three years.
The main goal/primary endpoint was to show fewer late‑stage (Stage III–IV) cancers in the Galleri arm on the logic that if Galleri works, some cancers that would have presented as Stage IV could be “shifted” to Stage II or III instead. That would obviously be good!
The trial did NOT achieve its primary endpoint: there was no statistically significant reduction in total Stage III–IV cancers in the Galleri arm vs control.
GRAIL emphasizes several secondary signals:
A “favorable trend” toward fewer Stage III–IV cancers within a pre‑specified group of 12 deadly cancers (but not statistically significant for the primary endpoint).
A substantial reduction in Stage IV diagnoses for those 12 deadly cancers—>20% fewer Stage IV cases in years two and three of screening—and similar trends across all cancers.
An increased detection of Stage I–II cancers in those deadly cancers in the Galleri arm.
Fewer cancers diagnosed through emergency presentations, which are linked to worse outcomes.
Test performance (PPV, specificity, CSO accuracy) was similar to previous North American trials; no major safety issues.
Independent experts that I read about quickly last night and this morning are more blunt:
STAT News and the New York Times both describe this as a failed trial in terms of its primary goal—Galleri did not significantly reduce late‑stage cancer diagnoses overall.
My Substack friend Dr. Eric Topol : “There’s no such thing in clinical trials as a favorable trend, especially in a trial that has got immense statistical power.”
Cancer researcher Dr. Adewole Adamson: “The study failed. End of story”.
Cancer researcher Dr. Richard Houlston calls Galleri “not a very good test” and says the results “don’t support rollout within the American health care system.”
So… we’re left with a nuanced picture, like always! Why is nothing simple? Nuance is ripe for exploitation by other people offering cheap and simple answers, so it’s important to chew on this for a while:
There may be meaningful stage shifting for some of the worst cancers (fewer Stage IV, more Stage I–II), but…
The trial was designed to show a clear reduction in overall Stage III–IV cancers and did not.
And we still have no mortality data—no proof yet that lives are actually being saved. That’s what we care the most about. That’s what earns a screening test a place in the primary care repertoire, and the United States Preventative Services Task Force (USPSTF) guidance for doctors and patients. Recall that RFK Jr. is quietly yet actively undermining the USPSTF as we speak. Recall what I said about opportunists swooping in to do the easy work of approving all sorts of vibe-driven tests and supplements and questionable treatments. Follow the money, and demand real expertise.
CancerGuard: the new competitor
A patient asked me just last week about a new test that was marketed to him after he received his routine colon cancer results from a Cologuard test (approved, and a good screening test for colon cancer that uses a stool sample). Should he pay the extra money to do this new blood test for multiple other types of cancer? Should I order it for him?
CancerGuard is another MCED test offered by the company who brings us Cologuard (Exact Sciences). It uses what called a multi‑omic approach:
DNA methylation (like Galleri, using technology that preserves DNA sequence)
Blood proteins associated with cancer.
The test is aimed at adults aged 50–84, and it screens for 50+ cancer types and subtypes—but excludes breast and prostate cancers (which are covered by other Exact Sciences products).
Key differences vs Galleri:
CancerGuard does not provide a specific cancer signal origin. A positive result triggers a broad imaging workup (which probably triggers a contrast CT of chest/abdomen/pelvis ± PET‑CT) rather than a more targeted search. That’s a lot of radiation, and potential to find other random stuff. See below.
It is cheaper—about $689 vs Galleri’s ~$949 list price.
It is considerably less validated in real‑world screening populations.
CancerGuard’s evidence
The headline numbers come from case‑control studies:
Development study: sensitivity 64.1%, specificity 97.4%.
Independent validation: sensitivity 55.6%, specificity 97.4%.
For six of the most lethal cancers (e.g., pancreatic, lung, ovarian), sensitivity 68%, and over one‑third of Stage I–II cancers detected.
However, all of this is from people already known to have cancer vs matched controls, not from large numbers of healthy people being screened. Exact Sciences explicitly warns that real‑world sensitivity may be lower than these case‑control estimates. The company is running additional prospective studies, but nothing at the scale of PATHFINDER 2 or NHS‑Galleri yet.
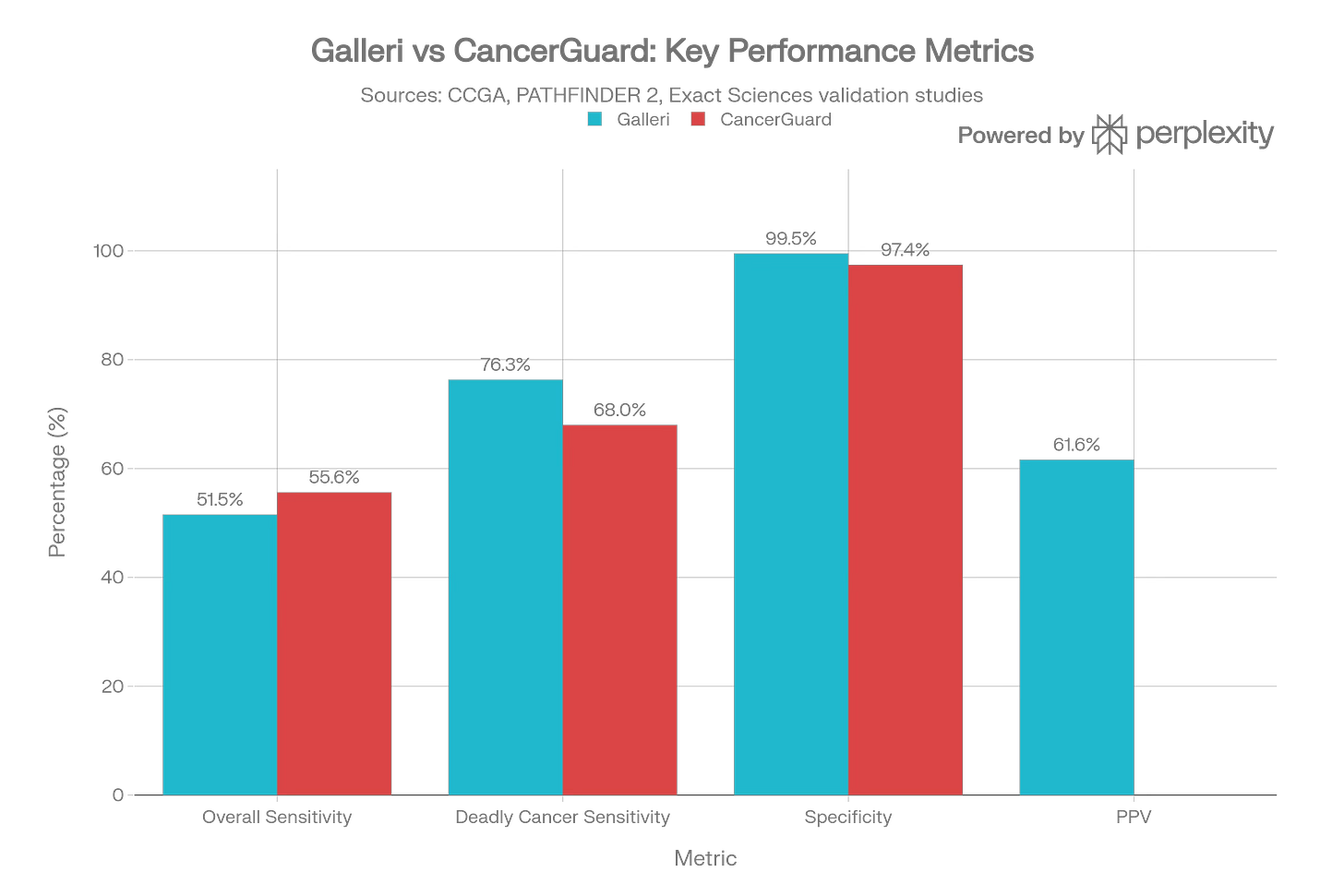
How do Galleri and CancerGuard compare?
Here’s a simplified comparison based on current data and manufacturer disclosures. Once again, I deployed Perplexity for help visualizing this data:
A few themes emerge to help us tease out the difference here:
Galleri is far better studied in real‑world screening; its strengths and weaknesses are clearer.
CancerGuard is cheaper and may ultimately benefit from multi‑omic design but currently lacks the same level of prospective validation.
Neither has shown mortality benefit, and Galleri has just failed that key trial meant to show a robust reduction in advanced cancers in the NHS.
Benefits: what these tests can actually offer right now
Despite the new negative trial, and my own reluctance to dive into all of this, I do want to acknowledge that there are real potential benefits to weigh.
Detecting cancers with no other screening tests
Both tests can pick up cancers like pancreatic, ovarian, liver, and some head and neck cancers, for which there are no routine screening tests for average‑risk people. Some of these are exactly the cancers we most fear, because they usually present late and are often lethal. Even catching a modest fraction of them earlier could matter for some individuals.
Very low false positive rates (especially Galleri)
Galleri’s specificity around 99.5–99.6% means very few people get a false alarm. That’s a major advantage over some traditional tests (like mammography, where 10% of screens can be called abnormal).
CancerGuard’s specificity (~97.4%) is somewhat lower but still high compared to many traditional screenings.
Helpful guidance when positive
Galleri’s Cancer Signal Origin prediction (correct >90% of the time) can help focus the diagnostic work‑up—targeted imaging rather than a whole‑body search. CancerGuard’s strategy is more broad‑brush (CT/PET‑CT), but its makers offer navigators and some financial support for follow‑up imaging. However, the financials of follow up testing are opaque to me, and are likely variable between insurance plans covering or not. It might all be out of pocket after the random screening test which was not indicated for chasing down a symptom or problem.
Signals of earlier detection in deadly cancers
Even in the NHS‑Galleri trial, which failed its primary endpoint, there appears to be:
*A reduction in Stage IV cancers, and
*A rise in Stage I–II cancers within the 12 deadliest cancers.
For most cancers, moving from Stage IV to Stage III can make a difference; for others, the gain is modest. Without full, disease‑by‑disease data and, crucially, survival data, we can’t yet quantify what this means in terms of lives saved.
Risks and limitations
Galleri’s sensitivity is lowest for Stage I cancers, catching maybe 1 in 6 at that earliest stage. That means a lot of true cancers are missed, especially early ones. CancerGuard’s case‑control data suggest better early‑stage performance, but we don’t yet know how that holds up in screening populations.
A negative MCED test therefore does not mean you’re cancer‑free. It simply means the test didn’t see a signal that day.
Real‑world reports show that some patients (and some telehealth companies) are treating Galleri as a kind of “all‑clear” when it’s negative, even delaying mammograms or colonoscopies. That’s dangerous. That was the outrage among people in the know who watched the Super Bowl ad. Given the modest sensitivity and the new NHS‑Galleri result, a negative MCED test should never be a reason to skip proven screening.
Are you an anxious person? That needs to be considered. A positive MCED test can launch weeks or months of:
Waiting for scans
Further testing and biopsies
Living with the worry: Is there cancer somewhere in me that we just haven’t found yet?
Some people wind up with a positive blood test but no detectable tumor, a situation one breast cancer researcher called the “Damocles syndrome”—a sword hanging over your head with no clear next step. Even if false positives are rare, each one is psychologically and financially costly.
So far, no MCED test has shown that its use reduces cancer deaths in a randomized trial. NHS‑Galleri was an important step, designed to show stage shift… but even that primary goal was not met. Demonstrating mortality benefit will require longer follow‑up and possibly different trial designs.
Without mortality data, earlier detection could still reflect lead‑time bias. This happens when we simply know about cancer being present longer without actually living longer.
Costs
For most of us non-billionaires, costs remain a consideration. Here is the best summary I could come up with, though prices often change and different sources quote different exact numbers:
Galleri costs roughly $949 (sometimes discounted to ~$700–$749 through certain providers or programs).
CancerGuard is priced around $689.
Most insurers and Medicare do not currently cover these tests.
In February 2026, the President signed the Nancy Gardner Sewell Medicare MCED Screening Coverage Act, which will eventually allow Medicare to cover FDA‑approved MCED tests that show clinical benefit—but not before around 2028, and likely with careful conditions. After the NHS‑Galleri result, experts are urging that coverage, if granted, be tied to clinical trials or registries so we keep learning rather than just paying.
So… should we get one?
At this point, no major medical society recommends routine MCED testing for everyone. The American Cancer Society’s guidance emphasizes shared decision‑making: a careful doctor‑patient conversation about what’s known, unknown, and what matters most to you personally.
Reasonable situations to consider an MCED test might include:
Being 50 or older
Having strong risk factors for cancers without standard screening (like significant family history, or a known hereditary cancer syndrome)
Understanding that the test is experimental, with no proven survival benefit
Not using a negative result as an excuse to skip mammograms, colonoscopies, or other recommended tests
Being able to afford the cost (and possible follow‑up costs) without giving up other important care
For many others, especially average‑risk adults, the most cautious and evidence‑driven stance is:
Focus on doing the proven screenings well and on time. Your good old-fashioned primary care doc is all about this.
Pay attention to symptoms and seek prompt evaluation
If you opt into an MCED test, treat it as participation in an evolving area of medicine—not as a guaranteed, life‑saving intervention.
A balanced way to explain this to patients
SO here is what I’m settling in with in terms of how I should answer my patients’ questions about those new blood tests for cancers. I’m thinking I might send them a link to this post, but some will prefer less of a deep dive. For them, perhaps I’ll say:
“These blood tests are a promising research frontier. They can sometimes catch cancers we’d otherwise miss, and they are quite good at avoiding false alarms.”
“However, they miss a lot of cancers, especially at the earliest stage, and the best large trial we have so far did not show a clear reduction in advanced cancers overall.”
“We also don’t yet know whether they help people live longer, and they can start a chain of follow‑up tests, cost, and anxiety.”
“If you’d like to consider one, we can talk through your personal risk, your values, and what you’d do with a positive or negative result. But whatever you decide, we should still keep up with regular mammograms, colon screening, Pap tests, and other proven tools.”
How does that sound?
For now, MCED tests sit in a gray zone: more than hype, but less than the revolution their marketing sometimes implies. They’re probably a glimpse of the future, but not yet the future fully realized.
As patients and clinicians, the most important thing is to keep two ideas in our heads at once:
The need for better early detection, especially for deadly cancers we currently find too late, is absolutely real.
The evidence we have on today’s MCED tests, especially after the NHS‑Galleri trial dropped yesterday, is not yet strong enough to treat them as a standard, life‑saving screening for everyone.
Ok, I’m done! I hope this deep dive was helpful. I relied heavily on sources like GRAIL, Exact Sciences, NY Times, STAT News, Perplexity, and other random sites found through an exhausting crunch of sources. Best of luck with your decisions, and please just consider this post one more educational input that informs an ultimate discussion with your doctor… who will not have time to go into the weeds like we just did together. That’s why I first started writing Examined in a quest to expand upon vital and overlooked ideas your family doctor might share — if only we had more time.
Enjoy your weekend if you can :) Significant snow predicted here in Philly tomorrow!




Ryan, congratulations on a great summary of these two "liquid cancer biopsies" and jumping so quickly on the Galleri NHS trial announcement. I'll wait for the publication in a journal before I weigh in on my Substack but it is fascinating how Grail headlined their PR announcement , "Landmark NHS-Galleri Trial Demonstrates a Substantial Reduction in Stage IV Cancer Diagnoses, Increased Stage I and II Detection of Deadly Cancers, and Four-Fold Higher Cancer Detection Rate" as excitingly positive whereas unbiased analysis interprets it as mostly negative.
It is the downstream testing that these screening tests generate that are the issue. A friend of mine had a positive Galleri for CRC. A colonoscopy followed and was normal and the GI doctor recommended one annually thereafter. Yikes!!!!!
I have come close to pulling the trigger on getting Galleri for myself but have held off due to concerns that I will be plunged into a miasma of anxiety and testing should it be falsely positive.
Doc Ryan,
As a retired Medical Technologist (with over 40 years of experience in urban major medical centers, one in particular that enrolled many pediatric oncology patients in trial studies) I nearly SWOONED reading this essay. I can't thank you enough for clearly explaining the sensitivity and specificity!
I could go on an on about how (IMHO) universal healthcare could not only catch cancer in early, treatable stages, but for diseases in particular! Working in the lab, as I'd release results into the computer system, I'd see the one line diagnosis associated with the patient. For the ER, sometimes what was there was the presenting complaint which was telling. I'd see such things as "couldn't afford my diabetes meds" or "rectal bleeding for five weeks" or such. I'd think about how much more this was going to cost ALL of us, as people without insurance will be treated (as they should be!) but the costs will be absorbed by those of us insured.
Very sadly, my sister passed from Primary Peritoneal Cancer. She had been having abdominal pain and I suggested she record her symptoms to bring with her. (Body awareness, just as mentioned in your last substack). Her doc suspected gallbladder and she was sent to a surgeon. The surgeon listened to her list of complaints and sent her for imaging, saying he "didn't want any surprises" when he operated. That's when the cancer was discovered. There is absolutely no early warning test for PPC that I am aware of, and as she had good insurance AND saw her doctor regularly, it would have been wonderful to have had this cancer caught early. But would it have helped? Much of her omentum was removed, and the doc said she had "hundreds of tumors like grains of sand", and she had chemo, but it just isn't a very treatable cancer.
I wish that the current administration would continue the Biden "cancer moon shot" as statistics show pretty clearly that either we ourselves will receive a cancer diagnosis in our lifetime OR someone close and dear to us will! Cancer starts at a molecular level and isn't diagnosed until it has grown large enough to be detected: by tumor imagining, by blood testing or, unfortunately, often by the impact of it causing symptoms. (The "warning signs") We need better treatments that cure without injuring the patient.
For those who swear that big pharma is in cahoots to keep from "curing" cancer as their drugs are big moneymakers, I highly recommend the Pulitzer Prize winning book "The Emperor of All Maladies: A biography of Cancer" by Dr. Siddhartha Mukherjee